Flukecides
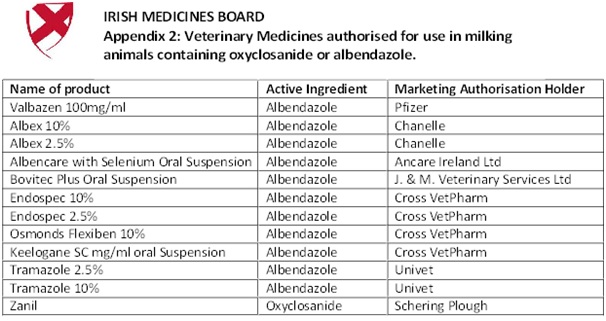
The IMB advises that flukicidal veterinary medicines containing oxyclosanide or albendazole, which already have a MRL established for milk, can continue to be used to treat adult fluke in animals which are intended for milk production. In these cases, the appropriate withdrawal periods indicated on the product labelling should be strictly followed. A list of products containing these substances is attached as Appendix 2.
Following an IMB initiative, maximum residue limits (MRLs) for milk in respect of the flukicidal substances clorsulon, closantel, nitroxinil, and triclabendazole were established by the European Commission in 2012. This has allowed some of the companies concerned to commission new residue studies in dairy animals producing milk for human consumption to establish appropriate withdrawal times. To date, the IMB has been provided with new milk residue depletion data for one authorised product containing triclabendazole as active substance (Fasinex 240 Oral Suspension for Cattle, VPA 10825/004/002) following administration of the product in dry-cows. A withdrawal period for use during the dry period has been established for this specific product. Note that this development is limited to this unique product; other triclabendazole products continue to be prohibited in animals producing milk for human consumption. However, it is possible that other companies might also conduct studies to support withdrawal periods for other individual products. It is also possible that new residue data to support an MRL for residues of rafoxanide in milk might also be developed, but this is not a matter for the IMB.
A recent European Commission Decision to harmonise the labelling warnings / withdrawal period recommendations in dry cows and pregnant heifers between products containing flukicidal substances, marketed in different EU Member States was published on 19th November 2012. This has resulted in a change to the existing prohibition on use of the products concerned in dairy cows and extended the prohibition to sheep and goats, throughout the EU area. The IMB is currently in the process of updating the marketing authorisations concerned to give effect to the labelling changes of products containing clorsulon, closantel, nitroxinil, triclabendazole or rafoxanide. It is expected that newly-labelled stock will be available in the second half of 2013. However, the net effect of this change is to bring Europe into line with the Irish position of 2010, as well as to give more precise instruction on use in maiden animals and in sheep and goats.
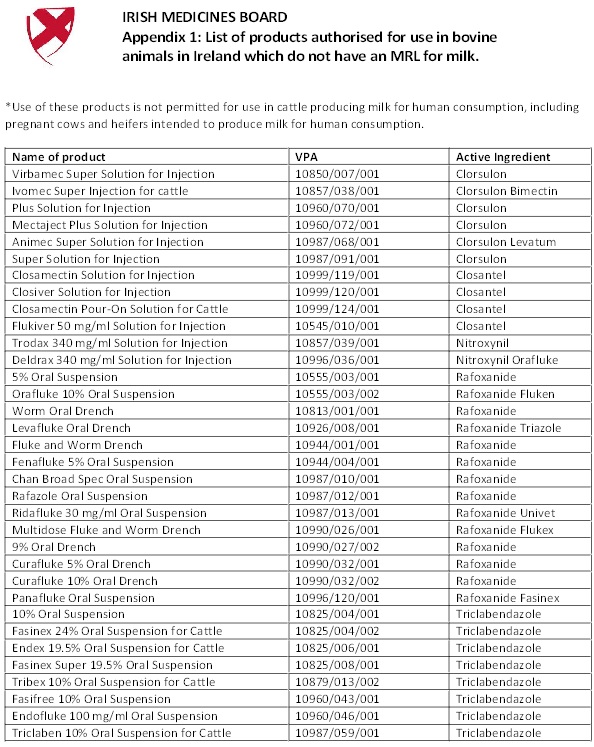
The IMB wishes to remind farmers of the ongoing restriction on the use of flukicides which do not have a maximum residue limit (MRLs - control thresholds for residues) for milk. As was the case in 2010, the Department of Agriculture continues to monitor milk for the presence of residues of flukicide drugs without MRL. The veterinary medicines concerned contain clorsulon, closantel, nitroxynil, rafoxanide and triclabendazole. Veterinary medicines containing these substances should not be used in dairy animals intended for milk production, including pregnant heifers intended for milk production for human consumption.
The labels of products containing clorsulon, closantel, nitroxynil, rafoxanide and triclabendazole have been updated during the course of 2010 to reflect the above warning. However, the labels of older stocks of veterinary medicines, which might be still in date and available on farms, might not be sufficiently explicit and apt in respect of the instruction not to use the products concerned in animals intended for milk production for human consumption. Farmers should follow the advice given here even where the label of the product available indicates otherwise. Note that the products concerned continue to be safe for use in animals which are not intended for use for milk production for human consumption, including beef suckler cows.
A list of products containing these substances which are affected by the IMB decision is attached as Appendix 1.